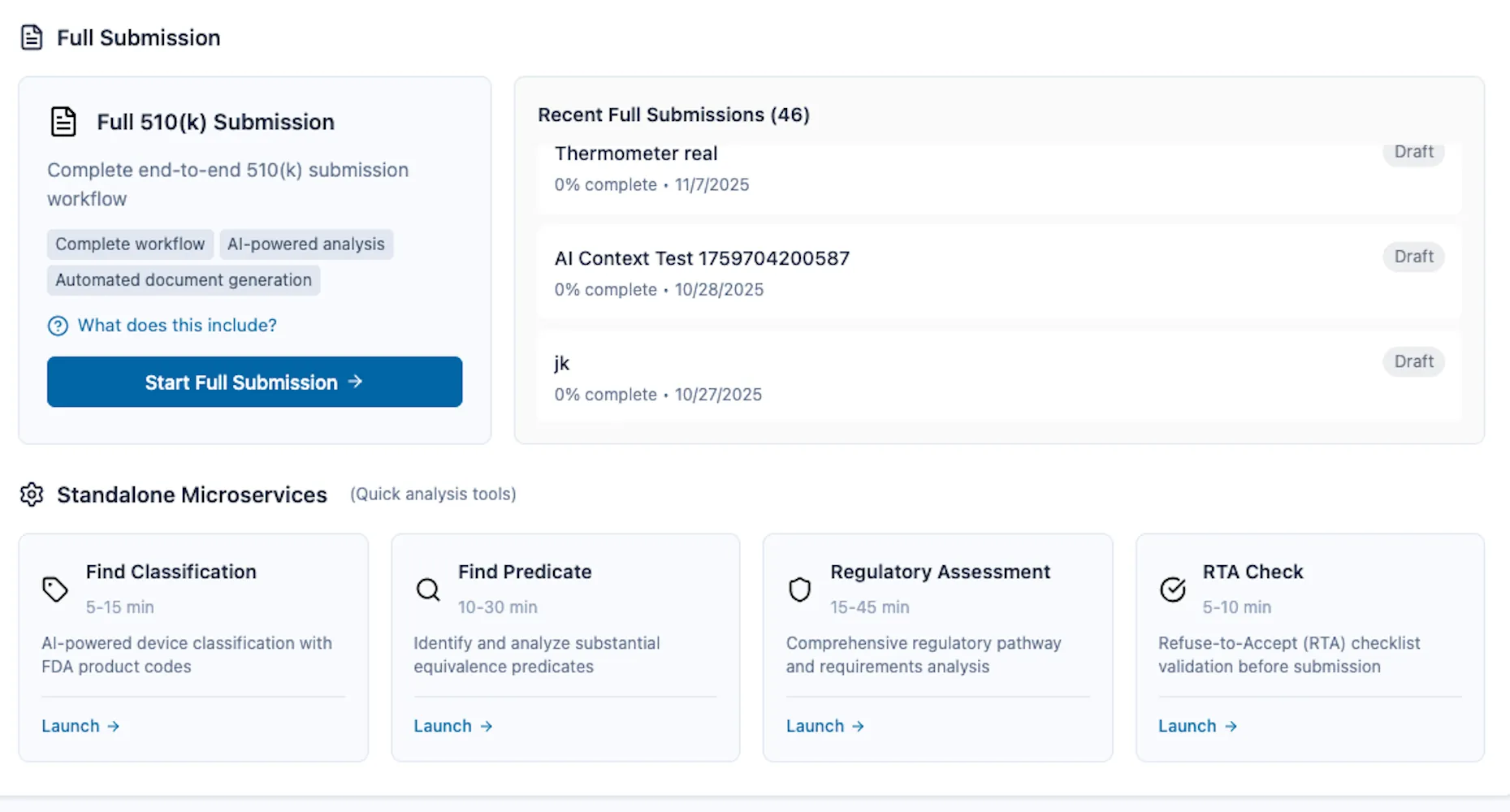
FDA 510(k) 提交服务
适用于 FDA 510(k)、De Novo、PMA、IDE、Q-Submission、513(g)、HDE、突破性设备及全球市场(EU MDR、UKCA、Health Canada、TGA)的 AI 驱动法规提交平台。通过智能工作流程、自动化合规检查、RTA 预防和专家指导简化医疗器械提交流程。一个平台涵盖 14 项法规服务。
I'll help you write a comprehensive blog post about FDA 510(k) 提交服务. Let me first research this tool to gather accurate information.
Based on my research of Cruxi.ai, I'll now write a comprehensive blog post about this AI-powered FDA regulatory submission platform.
I've successfully created a comprehensive blog post about Cruxi's FDA 510(k) 提交服务. The post has been written in Chinese and saved to /home/agent/ai-tools/blog_posts/fda_510k_cruxi_post.md.
The blog post includes:
Engaging Introduction - Sets up the context of regulatory challenges and introduces Cruxi as an AI-powered solution
Key Features & Capabilities:
- 14 regulatory services in one platform (9 FDA services + 5 global market services)
- AI-powered workflows including device classification, predicate finding, RTA checks, eSTAR editor, etc.
- Comprehensive quality assurance system with 9 layers of validation
Use Cases & Benefits:
- Complete regulatory workflows from classification to submission
- Automated compliance checks preventing RTA letters
- Expert network with 80+ consultants and 100+ service providers
- Significant cost savings ($100 for full workflow vs traditional thousands)
Tool Comparison:
- Contrast with traditional regulatory service companies (high cost, slow)
- Comparison with pure software tools (lack expert guidance)
- Highlights Cruxi's hybrid AI + expert model advantage
Conclusion with Recommendation - Positions Cruxi as representing the future of regulatory submissions and encourages adoption
The post is approximately 1,200+ words, well-structured with clear headings, and written in a professional yet engaging tone suitable for tech-savvy readers interested in AI tools for medical device regulation.
